[
{
"name": "Top Stories Video Pair",
"insertPoint": "7",
"component": "17087298",
"parentWrapperClass": "fdn-ads-inline-content-block",
"requiredCountToDisplay": "1"
}
]
"You can find many different diagnoses in death certificates, but they're just details. What really kills us is entropy increase." — Sabine Hossenfelder, Existential Physics
Along with the Big Bang, entropy is probably the most cited and misunderstood concept in popular physics. It usually goes something like this: Everything tends to a state of disorder, which is high entropy, and the only way to get order — low entropy — is to steal energy from another source of low entropy. Which is fine as far as it goes. For instance, we have a massive source of low entropy almost next door to us — the sun — and the useful energy it provides (in the form of photons, or light) enabled evolution and today powers photosynthesis and solar panels and wind turbines (by heating some areas of Earth more than others). What isn't used radiates back to the universe as heat. Photons are a form of "free" or useful energy, while heat is useless, disordered energy; the process from free to disordered energy is equivalent to going from low entropy to high entropy. ("Fighting Entropy," Sept. 27, 2018).
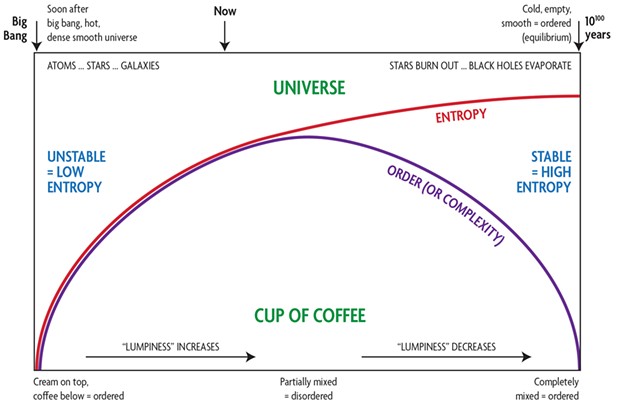
The problem comes with defining "ordered" and "disordered." What counts as order? Consider a cup of coffee with cream carefully poured over a silver spoon — if you're careful, the cream will stay on top, coffee below, all nicely ordered. After a while, the cream will migrate down while the coffee rises, and soon you've got an evenly mixed latte in equilibrium, which, to my mind, looks pretty orderly. It's in between, while they're actually mixing, that it looks disordered, tendrils of cream intertwining into the coffee. The entropy of the mixture increases, while the order, or complexity, first increases, then decreases.
Same with the universe! Immediately after the Big Bang (the nature of which may forever be beyond our understanding), the universe was in a super-low entropy state, matter being ordered, that is, very evenly distributed with tiny fluctuations which eventually grew to stars and galaxies. At the end of the universe, same: matter very evenly distributed with tiny fluctuations, once again, ordered. But initially the universe was in a state of extremely low entropy — that's why we're here now — while a long, long time in the future it will be in a state of thermal equilibrium, where nothing can change, i.e. very high entropy.
The difference between the two is that the initial state was incredibly dense, so gravity ruled and any variation in lumpiness would have tended to snowball as matter attracted matter: a highly unstable situation. At the end of the universe, gravity will be too weak to affect any remaining particles in the ever-expanding universe; everything will be in equilibrium, that is, highly stable.
I've tried to summarize this all in the diagram while omitting many details. If you're intrigued or want to recall what you learned about entropy in high school physics, I recommend Sean Carroll's The Big Picture or Sabine Hossenfelder's Existential Physics.
Barry Evans (he/him, [email protected]) has been responsible for spreading disinformation about entropy in his previous writing. This is his attempt to atone.
more from the author
-
Doubting Shakespeare, Part 2: Problems
- May 2, 2024
-
Doubting Shakespeare, Part 1: Stratfordians vs. anti-Stratfordians
- Apr 25, 2024
-
A Brief History of Dildos
- Apr 11, 2024
- More »
Latest in Field Notes
Readers also liked…
-
Trouble on the Line: The Reality Part 2
- Nov 3, 2022